Ustekinumab Biosimilar
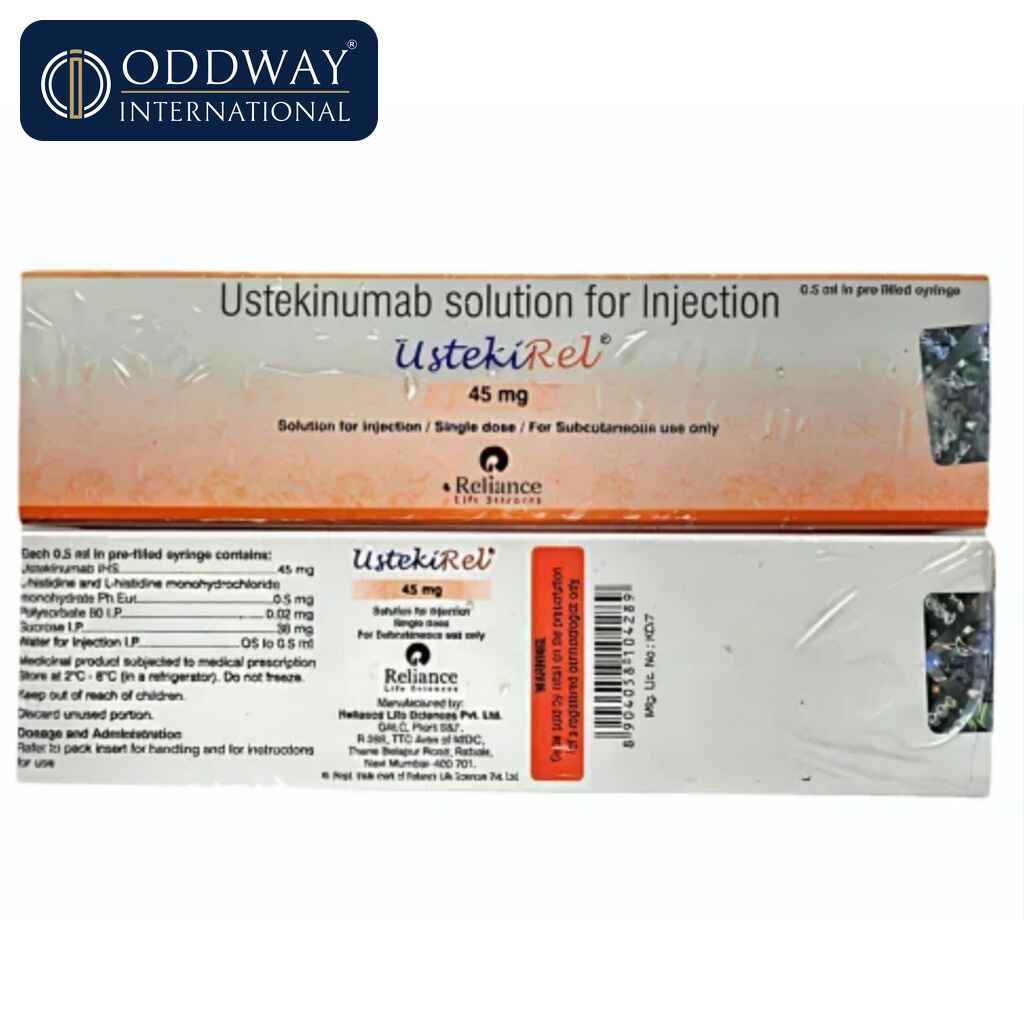
Product Name: UstekiRel
Composition: Ustekinumab (biosimilar)
Dosage Form: Injection (parenteral)
Strength: 45mg/90mg
Manufacturer: Reliance Lifesciences
Send Enquiry for Ustekinumab Biosimilar
Product overview for Ustekinumab Biosimilar supplier requirements
Ustekinumab Biosimilar is a specialty biologic supplied for institutional procurement programs, distributor networks, and cross-border tenders where permitted. Oddway International supports B2B buyers who require consistent sourcing, controlled handling, and documentation alignment for regulated and semi-regulated markets. Moreover, our team coordinates brand preferences (subject to availability), batch planning, and shipment scheduling to help you maintain continuity in your supply chain.
As an export-focused partner, we prioritize clear communication on lead times, shelf-life expectations, and country-specific import needs. Additionally, we support procurement teams with pre-shipment document review to reduce customs queries and clearance delays. However, final import approval always remains subject to local regulations and the buyer’s licensing requirements.
Therapeutic category/Drug classification
Ustekinumab Biosimilar is classified as a monoclonal antibody (biosimilar) within the broader category of immunology biologics. It is typically handled as a temperature-sensitive specialty product depending on the manufacturer’s labeling and stability profile. Therefore, buyers should confirm local registration status, pharmacovigilance obligations, and handling requirements before placing an order.
Ustekinumab Biosimilar bulk pricing & minimum order quantity (MOQ) structure
Oddway International provides structured quotations for institutional and trade customers seeking Ustekinumab Biosimilar wholesale procurement. Pricing is influenced by requested brand/manufacturer, strength and presentation, destination country requirements, and shipment mode (air/express or consolidated freight). Consequently, we recommend sharing your target market, preferred Incoterms, and required documentation set so we can quote accurately.
- MOQ: Typically aligned to manufacturer shipper units and cold-chain feasibility (subject to availability).
- Tiered volumes: Better commercial terms may apply for repeat schedules and consolidated orders.
- Quotation format: Proforma invoice with batch/lead-time guidance where available.
- Commercial clarity: We support requests for indicative Ustekinumab Biosimilar price ranges for budgeting, subject to final confirmation.
For tender-like requirements, we can align supply planning with periodic deliveries, where permitted, to support uninterrupted institutional demand. Moreover, we can discuss alternatives if a specific brand faces allocation or longer lead times.
Available strengths, packaging & Ustekinumab Biosimilar regulatory compliance
Ustekinumab Biosimilar may be available in multiple strengths and presentations depending on the manufacturer and destination market. Packaging formats can vary by brand and may include different device configurations. Therefore, Oddway International confirms the exact strength, pack presentation, labeling language, and storage instructions before dispatch.
We support regulatory alignment by providing documentation typically requested in B2B trade, such as Certificate of Analysis (COA) where available, batch details, and export documentation support. Additionally, we coordinate on country-specific needs such as legalized paperwork or importer-specific declarations, subject to local regulations and manufacturer availability.
Wholesale, export & named patient supply capabilities for bulk purchase Ustekinumab Biosimilar
Oddway International supports routine wholesale export as well as urgent access pathways. If you manage special access programs, we can assist through named patient supply workflows where permitted, including tight timelines and document coordination. Furthermore, we work with hospitals, specialty clinics, and procurement agents to align on patient-specific documentation requirements without making patient-facing claims.
For broader sourcing needs across biologics, buyers can also engage us as a biosimilar drugs supplier to streamline vendor management and consolidate shipments when feasible. As a result, procurement teams can reduce administrative load while maintaining traceability and compliance expectations.
Quality assurance & global standards for Ustekinumab Biosimilar
We follow a quality-first approach suitable for international trade channels. This includes supplier qualification, batch and expiry verification, and careful coordination of storage and dispatch conditions. Moreover, we support Good Distribution Practice (GDP)-aligned processes through appropriate packaging selection and lane assessment, especially for temperature-sensitive products.
Where required, we can share available quality documents such as COA, MSDS/SDS, and shipping condition notes. However, document availability can vary by manufacturer and market channel. Consequently, we confirm the document set at quotation stage to match your importer and customs expectations.
Why source Ustekinumab Biosimilar from Oddway International
Oddway International is a trusted Indian pharmaceutical exporter supporting regulated and semi-regulated markets with responsive coordination and transparent order handling. If your procurement team needs a reliable Ustekinumab Biosimilar supplier for repeat cycles, we help you plan shipments, manage lead times, and standardize documentation across orders. Additionally, we provide professional communication across time zones to keep your internal stakeholders updated.
For broader specialty portfolios, many buyers also work with us as a specialty drugs supplier to simplify sourcing across complex therapies and special handling requirements. Therefore, you can centralize procurement discussions while maintaining product-by-product compliance.
International logistics & documentation Support for international medicine shipping
We manage export packing, temperature-control planning where applicable, and courier/air-freight options based on urgency and destination constraints. Additionally, we assist with standard export documentation such as commercial invoice, packing list, airway bill support, certificate of origin where applicable, and any buyer-requested documents subject to feasibility.
To support lane planning and customs readiness, buyers can review our capabilities for international medicine shipping. Consequently, procurement teams gain clearer expectations on timelines, tracking, and delivery coordination for hospital, distributor, or institutional receiving points.
5 reviews for Ustekinumab Biosimilar
Shipping Options
We offer a variety of shipping options, including Express Mail Service (EMS), USPS, DHL, FedEx, TNT, UPS, Aramex, Air Cargo, and sea freight.
Terms and Conditions
- Bulk Orders: You are responsible for any local import duties and taxes in your country.
- For Patients: When ordering prescription medicines for personal use, or for a friend or relative, a valid medical practitioner's script or prescription is required.
- Return and Refund Policy: Due to the nature of our products, we cannot accept returns or exchanges once a purchase is made. However, in the event of non-delivery, you are eligible for either a 100% refund or a reshipment of your order.
Frequently Asked Questions
Share your required strength/presentation (or preferred brand), destination country, target quantity, and importer details. We will confirm availability, lead time, MOQ, and provide a proforma invoice with the feasible documentation set, subject to local regulations.
Typical documentation includes commercial invoice, packing list, airway bill support, and certificate of origin where applicable. COA and MSDS/SDS can be provided where available from the manufacturer/channel. Final documents depend on destination requirements and feasibility.
Yes, where permitted, we can coordinate Named Patient Supply orders with urgent timelines, patient-specific paperwork routing, and controlled shipping planning. Requirements vary by country, so we align the process with the buyer’s hospital/physician documentation and import rules.
MOQ is generally based on manufacturer shipper units and handling constraints (including temperature-control feasibility). Bulk purchase orders are supported, and we can discuss tiered volumes and repeat supply schedules depending on availability.
We support express courier and air-freight options depending on destination, urgency, and product handling needs. For temperature-sensitive shipments, we plan appropriate packaging and routing. Delivery timelines and serviceability remain subject to carrier and customs conditions.

Hassan Al-Farsi –
Oddway coordinated an urgent shipment and kept us informed at each step. Documentation was prepared neatly and the delivery reached within the expected window.
Anika van Dijk –
Professional handling and quick responses on email. We appreciated the structured proforma invoice and the way they confirmed packaging and storage notes before dispatch.
Samuel Okoye –
Good experience overall. They shared realistic lead times, provided the paperwork we requested, and coordinated delivery details with our clearing agent without back-and-forth.
Laura Mendes –
We had tight internal timelines and Oddway kept updates consistent. A couple of items needed reconfirmation on our side, but their team stayed patient and organized.
Imran Qureshi –
Clear communication from quotation to dispatch. They double-checked our document list before shipping, which helped customs clearance go smoothly.