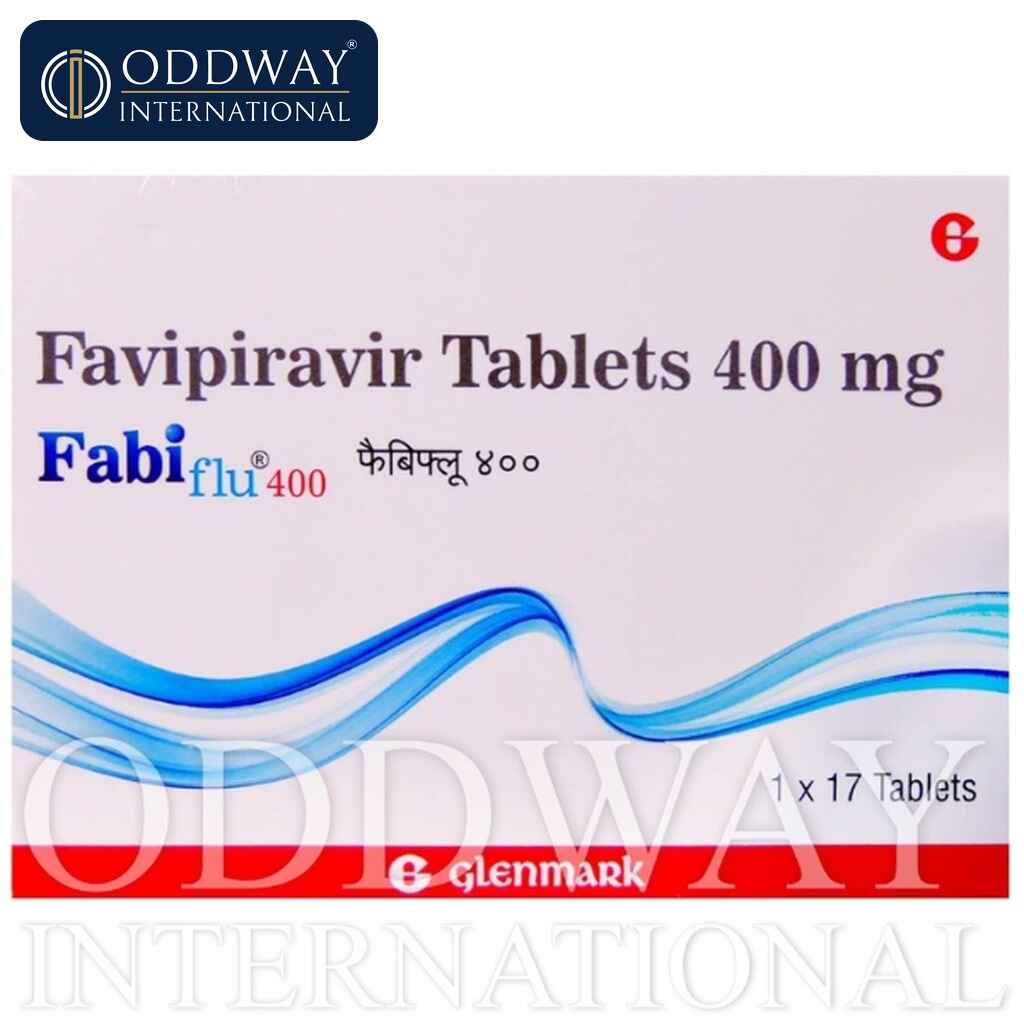
Favipiravir Tablets 200/400 mg
Composition: Favipiravir 400mg
Strength Available : 200 mg & 400 mg
Form: Tablet
Packing: Pack of 17 Tablets
Country Of Origin: India
Favipiravir Brand Name :
- Fabiflu 400mg/200mg (Manufactured By : Glenmark Pharmaceuticals)
- Favivir (Manufactured By : Hetero Drugs Limited)
Send Enquiry for Favipiravir Tablets 200/400 mg
Product overview for Favipiravir Tablets 200/400 mg wholesale
Favipiravir Tablets 200/400 mg are supplied for institutional procurement and cross-border trade programs where permitted and subject to local regulations. Oddway International supports importers, distributors, and hospital buyers with compliant sourcing pathways, export-ready documentation, and shipment planning aligned with destination-country requirements. Consequently, buyers can consolidate procurement, reduce vendor complexity, and maintain continuity across tenders and recurring purchase cycles.
As an India-based exporter serving regulated and semi-regulated markets, we coordinate product selection, batch planning, and dispatch timelines with clear lead-time communication. Moreover, we support B2B buyers who require manufacturer documentation, controlled packing practices, and professional coordination for urgent allocations or time-sensitive orders.
Therapeutic category/Drug classification
Favipiravir is classified under antiviral agents. Procurement teams typically manage it within anti-infective/antiviral portfolios for institutional supply, subject to country-specific approvals and protocols. However, Oddway International does not provide medical advice and supplies only in accordance with applicable laws, import permits, and local regulatory frameworks.
Favipiravir Tablets 200/400 mg bulk pricing & minimum order quantity (MOQ) structure
Oddway International offers structured quotes for bulk purchase Favipiravir Tablets 200/400 mg based on strength, packaging configuration, destination market requirements, and documentation scope. Therefore, we propose the most suitable MOQ and price tier after confirming the target country, required shelf-life, and any labeling or language needs.
- MOQ options: typically aligned to shipper cartons/master cases; institutional MOQs available on request
- Price drivers: strength (200 mg/400 mg), batch availability, requested manufacturer, and compliance documents
- Quote format: ex-works/fob/cif where applicable, with lead time and validity clearly stated
- Order planning: repeat-order schedules supported for distributors and hospital networks
Available strengths, packaging & Favipiravir Tablets 200/400 mg exporter regulatory compliance
Favipiravir Tablets are commonly requested in 200 mg and 400 mg strengths. Packaging can vary by manufacturer and destination requirements; we confirm pack size, outer carton configuration, and labeling details during the proforma stage. Additionally, we support documentation packages aligned to the importing country’s needs, including batch-specific paperwork and export documents.
Regulatory support can include, where available and applicable: Certificate of Analysis (COA), manufacturing licenses, Free Sale Certificate/CPP (as applicable), and stability/shelf-life confirmation. Consequently, procurement teams can evaluate compliance readiness before dispatch and align internal QA release processes.
Key advantages for importers, distributors & hospitals using a Favipiravir Tablets 200/400 mg supplier
Oddway International operates as a B2B-focused partner for repeat procurement, tender participation support, and institutional supply coordination. Moreover, we help buyers reduce delays by validating documentation requirements early and matching the supply plan to the destination’s clearance process.
- Clear batch and shelf-life confirmation before shipment
- Documentation coordination for customs and internal QA review
- Consolidation options with other molecules to optimize freight
- Responsive communication across procurement, QA, and logistics teams
Wholesale, export & named patient supply capabilities with Favipiravir Tablets 200/400 mg wholesaler
We support wholesale export programs for licensed importers as well as urgent Named Patient Supply (NPS) requests where permitted. Furthermore, for NPS, we coordinate product identification, documentation alignment, and expedited dispatch planning to meet time-sensitive needs without compromising compliance.
For broader portfolio sourcing, buyers can also consolidate with generic drugs and related categories, depending on local registration status and import rules. As a result, procurement teams can streamline vendor management and shipping schedules.
Quality assurance & global standards for Favipiravir Tablets 200/400 mg supplier programs
Oddway International follows structured QA coordination practices suitable for international trade. We facilitate batch documentation, verify pack integrity expectations, and support GDP-aligned handling practices where required. Additionally, we can share SDS/MSDS where applicable and coordinate special handling instructions with freight partners.
Where permitted, we also support institutional buyers procuring through Patient access medicine pathways for urgent cases, ensuring the process remains documentation-driven and compliant with local requirements.
Why source Favipiravir Tablets 200/400 mg from Oddway International
Oddway International is a trusted Indian pharmaceutical exorter supporting buyers across multiple regions with consistent communication, export-ready paperwork, and supplier coordination. Moreover, we work with subject-to-availability / requested brand options so procurement teams can select suitable manufacturers based on market preference and documentation needs.
We prioritize clarity on lead times, batch allocation, and shipment milestones. Consequently, importers and hospitals can plan receiving schedules, internal QC release, and downstream distribution with fewer surprises.
International logistics & documentation Support for international medicine shipping
Oddway International coordinates international medicine shipping with export documentation support suitable for commercial and institutional consignments. We assist with invoice/packing list preparation, HS code guidance where appropriate, and courier/air-freight planning based on urgency and destination clearance norms.
Additionally, buyers sourcing broader anti-infective portfolios can align Favipiravir procurement with Antiviral drugs category requirements to standardize documentation and shipping workflows across products, subject to local regulations.
10 reviews for Favipiravir Tablets 200/400 mg
Shipping Options
We offer a variety of shipping options, including Express Mail Service (EMS), USPS, DHL, FedEx, TNT, UPS, Aramex, Air Cargo, and sea freight.
Terms and Conditions
- Bulk Orders: You are responsible for any local import duties and taxes in your country.
- For Patients: When ordering prescription medicines for personal use, or for a friend or relative, a valid medical practitioner's script or prescription is required.
- Return and Refund Policy: Due to the nature of our products, we cannot accept returns or exchanges once a purchase is made. However, in the event of non-delivery, you are eligible for either a 100% refund or a reshipment of your order.
Frequently Asked Questions
Yes. We support B2B procurement for licensed importers, distributors, and hospitals, subject to local regulations. We share availability, lead time, and documentation scope before order confirmation.
Documentation may include commercial invoice, packing list, COA, and other manufacturer/regulatory papers where applicable and available. The exact set depends on destination requirements and the requested brand.
Where permitted, we can support NPS requests by coordinating product selection, paperwork alignment, and expedited shipping options. Final supply depends on local rules and case documentation.
MOQ typically aligns with shipper cartons/master cases and varies by strength, manufacturer, and destination. Share your target country and preferred strength to receive a tailored MOQ and quote.
We can arrange courier or air-freight options depending on urgency, destination clearance norms, and shipment size. We coordinate export packing and documentation to support smooth customs processing.


Ravi Narang –
We consolidated this order with other items and it reduced our freight complexity. The team provided timely confirmations on batch and dispatch, and the documents were organized and easy for our QA review.
Sophie Laurent –
We needed an urgent, documentation-driven shipment. They were responsive and kept the process moving without overpromising. Good coordination with the courier partner.
Carlos Mendez –
Oddway coordinated the paperwork and packing details well. What helped most was the proactive updates—our receiving team knew exactly what to expect for clearance and delivery.
Lina K. –
We had a few labeling questions for our market, and the team handled them quickly with the manufacturer. Overall process was smooth and professional.
Omar Al-Mansouri –
Communication was clear from quotation to dispatch. Documents were shared in advance for our internal checks, and the shipment timeline matched what was agreed.
Cameron Green –
Their fair pricing feels like a stand against overpriced healthcare everywhere else.
Ashley –
I thought quality healthcare was out of reach, but this pharmacy proved otherwise.
Ashton –
Ordering takes minutes, and delivery is lightning-fast.
Devon Conway –
Ordering here feels like stepping into a futuristic pharmacy, where everything works flawlessly and stress-free.
Bellarine –
Their team explained complex medical terms in simple language, making me feel confident in my choices.