Cyclophosphamide injection
Product Name: Cyclophosphamide injection
Composition: Cyclophosphamide
Dosage Form: Injection (IV use; reconstitution/infusion as applicable)
Strength: 500 mg/1000mg
Packaging: Vials
IV Cyclophosphamide brand:
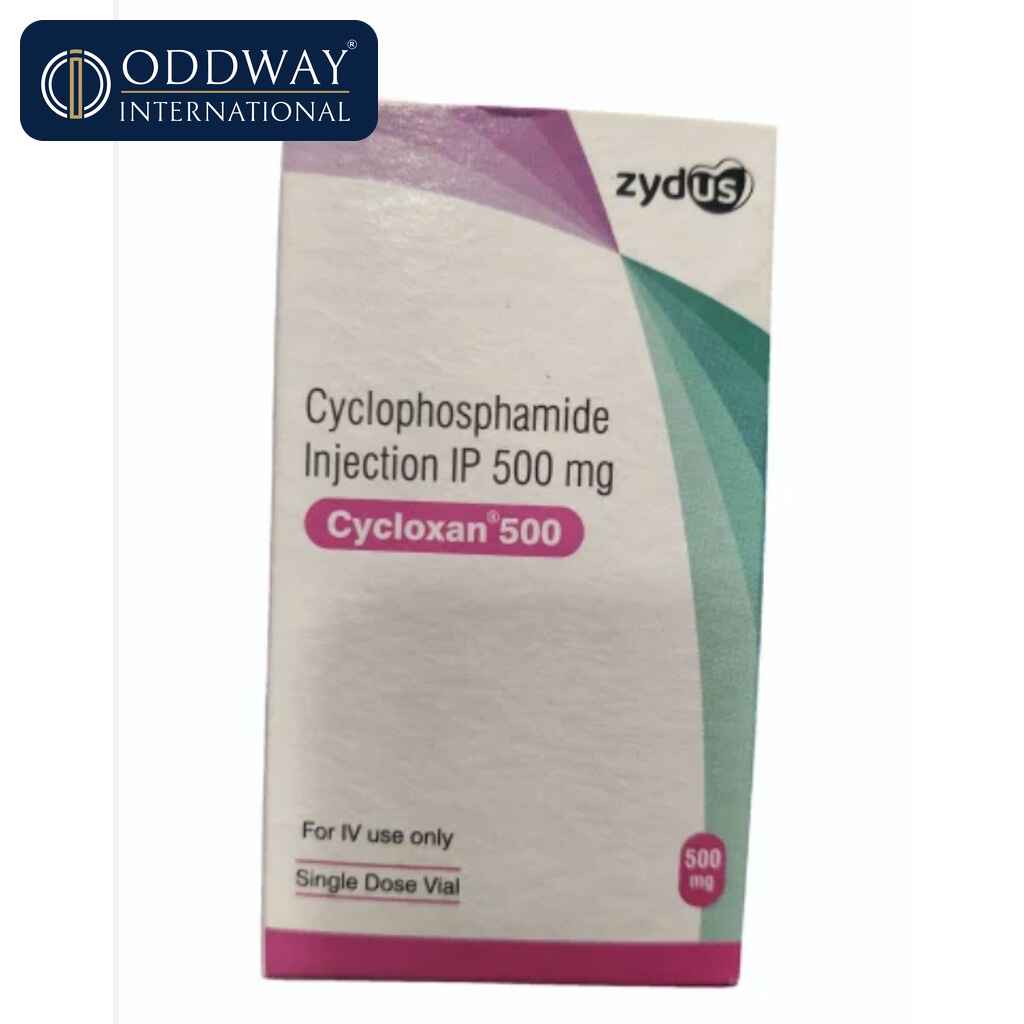
Cycloxan (Zydus)
Cyphos (Getwell Lifesciences)
Cycram 500mg Injection (Vhb Life Sciences)
Send Enquiry for Cyclophosphamide injection
Product overview for Cyclophosphamide injection wholesale
Cyclophosphamide injection is a sterile parenteral formulation widely procured by hospitals, oncology centers, government tenders, and institutional buyers for controlled distribution channels. Oddway International supports B2B procurement with an export-focused supply model designed for regulated and semi-regulated markets, where permitted and subject to local regulations. We coordinate brand selection, documentation readiness, and shipment planning to match importer requirements and timelines.
As an India-based supplier to Myanmar, Egypt, Angola, Afghanistan, Ghana, Kenya, Tajikistan, Venezuela, Cameroon, and Madagascar etc, we prioritize traceable sourcing, clear batch-level paperwork, and responsive coordination for repeat orders. Additionally, we align deliveries with your receiving SOPs, including temperature considerations, handling instructions, and customs documentation. Consequently, procurement teams can reduce lead-time uncertainty and maintain continuity of supply for institutional demand planning.
Therapeutic category/Drug classification
Cyclophosphamide is classified as an antineoplastic and immunosuppressive agent (alkylating agent). It is typically handled as a specialty injectable within hospital supply chains. However, product selection, labeling, and import permissions vary by country; therefore, buyers should confirm local regulatory requirements and tender specifications prior to purchase.
Cyclophosphamide injection bulk pricing & minimum order quantity (MOQ) structure
For B2B procurement, we provide structured quotations that reflect strength, brand/manufacturer preference, destination country, and required documentation. While buyers often request Cyclophosphamide injection price comparisons across brands, we focus on total landed-cost drivers such as freight mode, insurance, and clearance needs. Moreover, we can align quotes to tender formats or proforma invoice templates used by your institution.
MOQ typically depends on the selected presentation and availability from the requested brand. For example, consolidated orders across multiple oncology injectables can reduce per-line MOQ impact and improve freight efficiency. As a result, wholesalers and hospital buyers can plan mixed-carton shipments while maintaining compliant packing and labeling, subject to local regulations.
Available strengths, packaging & regulatory compliance for Cyclophosphamide iv infusion supplier
Oddway International can source Cyclophosphamide injection in commonly traded strengths and vial presentations, subject to availability and market authorization. Packaging options may include single vials and secondary packs suitable for institutional supply channels. Additionally, we support requests for specific labeling needs (e.g., English-only packs) where feasible and permitted by the manufacturer and regulations.
Regulatory support may include batch-specific documentation such as Certificate of Analysis (COA), invoice/packing list alignment, and other quality documents requested by importers. However, final acceptance remains subject to your local authority requirements and the product’s registration status in the destination market.
- Strength options: subject to availability and requested brand
- Dosage form: sterile injection for IV administration as applicable
- Documentation support: COA and shipment paperwork on request
- Compliance approach: export-ready coordination per destination needs
Wholesale, export & named patient supply capabilities for Cyclophosphamide injection exporter
We support Cyclophosphamide injection exporter requirements for wholesalers, distributors, and institutional buyers, including repeat supply planning and urgent line-item fulfillment. Furthermore, we can coordinate special handling instructions and route planning to minimize transit risk, especially for time-sensitive hospital procurement cycles.
For patient-specific access programs, we also support named patient supply workflows where permitted. This includes document coordination and shipment prioritization based on consignee requirements, while maintaining a professional, compliant B2B process and avoiding patient-facing claims.
Quality assurance & global standards aligned with bulk purchase iv Cyclophosphamide
Quality assurance remains central for any bulk purchase iv Cyclophosphamide order. We emphasize batch traceability, document completeness, and packaging integrity checks prior to dispatch. Additionally, our team supports GDP-aligned logistics practices and can share available quality documents such as COA and, where applicable, SDS/MSDS upon request.
Moreover, we coordinate export packing suitable for international transit and help reduce discrepancies that can delay customs clearance. Consequently, procurement teams receive consistent paperwork and predictable shipment preparation aligned with institutional receiving standards.
Why source Cyclophosphamide injection from Oddway International
Oddway International operates as a trusted India-based exporter serving global buyers with responsive communication and practical procurement support. If you are evaluating a reliable pharmaceutical exorter for oncology injectables, we provide structured quotations, brand options (subject to availability), and coordination across documentation and dispatch milestones.
Additionally, we support consolidated procurement across multiple product lines, which helps wholesalers and hospitals optimize freight costs and reduce repeated clearance work. Therefore, buyers benefit from a single point of coordination for export documentation, packing, and shipment tracking.
International logistics & documentation Support for Cyclophosphamide injection wholesale
We manage end-to-end export coordination including proforma invoice, commercial invoice, packing list, and other documents typically requested by importers. Furthermore, we can support courier and air freight planning for urgent needs, as well as standard air/sea options for routine procurement. For routing and delivery planning, buyers can also reference our international medicine shipping capabilities.
However, customs requirements differ by destination; therefore, we ask buyers to share import license details and consignee preferences early in the process. As a result, we can align labeling, documentation, and dispatch timelines to reduce clearance delays and ensure smooth receipt at the destination warehouse or hospital store.
5 reviews for Cyclophosphamide injection
Shipping Options
We offer a variety of shipping options, including Express Mail Service (EMS), USPS, DHL, FedEx, TNT, UPS, Aramex, Air Cargo, and sea freight.
Terms and Conditions
- Bulk Orders: You are responsible for any local import duties and taxes in your country.
- For Patients: When ordering prescription medicines for personal use, or for a friend or relative, a valid medical practitioner's script or prescription is required.
- Return and Refund Policy: Due to the nature of our products, we cannot accept returns or exchanges once a purchase is made. However, in the event of non-delivery, you are eligible for either a 100% refund or a reshipment of your order.
Frequently Asked Questions
Yes. We support B2B procurement for wholesalers, distributors, hospitals, and institutional buyers, subject to product availability and destination-country regulations. We provide export-ready quotations and coordinate required documents.
Commonly requested documents include commercial invoice, packing list, and batch-related documents such as COA (on request). Additional documents can be arranged based on importer and customs requirements, subject to feasibility.
Where permitted, we can coordinate Named Patient Supply workflows, including shipment prioritization and documentation alignment with consignee requirements. Final acceptance depends on local rules and the receiving institution’s process.
MOQ depends on the requested strength, presentation, and brand/manufacturer availability. We can also plan consolidated orders across multiple items to improve freight efficiency and reduce per-item MOQ impact.
We can arrange courier or air freight for urgent requirements and standard air/sea freight for routine procurement, depending on destination and importer preferences. We coordinate packing, labeling checks, and shipment tracking support.


Jonas P. –
Reliable export support and responsive email communication. We received the batch documents we requested and the shipment arrived within the expected window.
Mariana L. –
Good coordination for a mixed order. The team helped us consolidate multiple line items so we could optimize freight without delaying the shipment.
Ahmed K. –
Professional handling and consistent updates. What we appreciated most was the alignment of invoice/packing list details with our internal receiving format, which reduced back-and-forth.
Sofia R. –
We had a tight timeline and Oddway coordinated the shipment milestones well. The team followed up quickly when customs asked for an additional clarification.
Daniel M. –
Clear communication from quotation to dispatch. Documents matched our import requirements and the packing was done neatly for receiving.