

Shipping Options
We offer a variety of shipping options, including Express Mail Service (EMS), USPS, DHL, FedEx, TNT, UPS, Aramex, Air Cargo, and sea freight.
Terms and Conditions
- Bulk Orders: You are responsible for any local import duties and taxes in your country.
- For Patients: When ordering prescription medicines for personal use, or for a friend or relative, a valid medical practitioner's script or prescription is required.
- Return and Refund Policy: Due to the nature of our products, we cannot accept returns or exchanges once a purchase is made. However, in the event of non-delivery, you are eligible for either a 100% refund or a reshipment of your order.

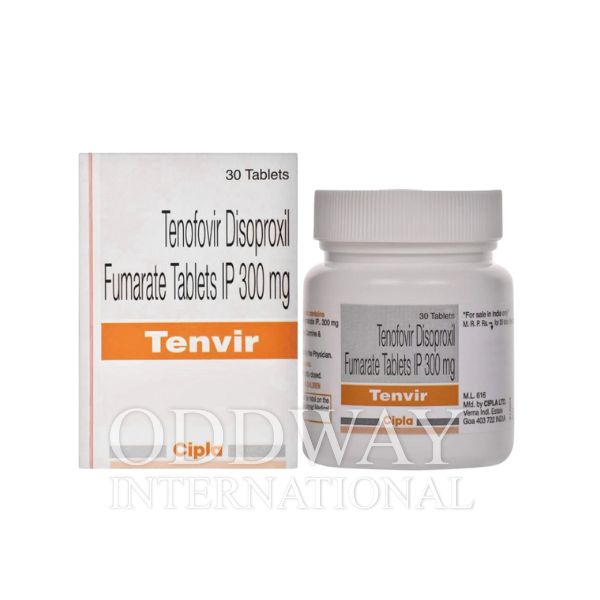





Megan Schutt –
The website offers product descriptions in multiple languages, which makes it much easier for international customers to understand what they’re buying.
Sophie Molineux –
The payment system was smooth, with a wide range of secure methods.
Nathan McSweeney –
I’ve never seen such a meticulous approach to packaging.
Derek Pringle –
I love their focus on building trust. Everything about their process, from order to delivery, feels customer-focused and reliable.
Jonathan Powell –
The pharmacy’s transparency about shipping timelines was refreshing.