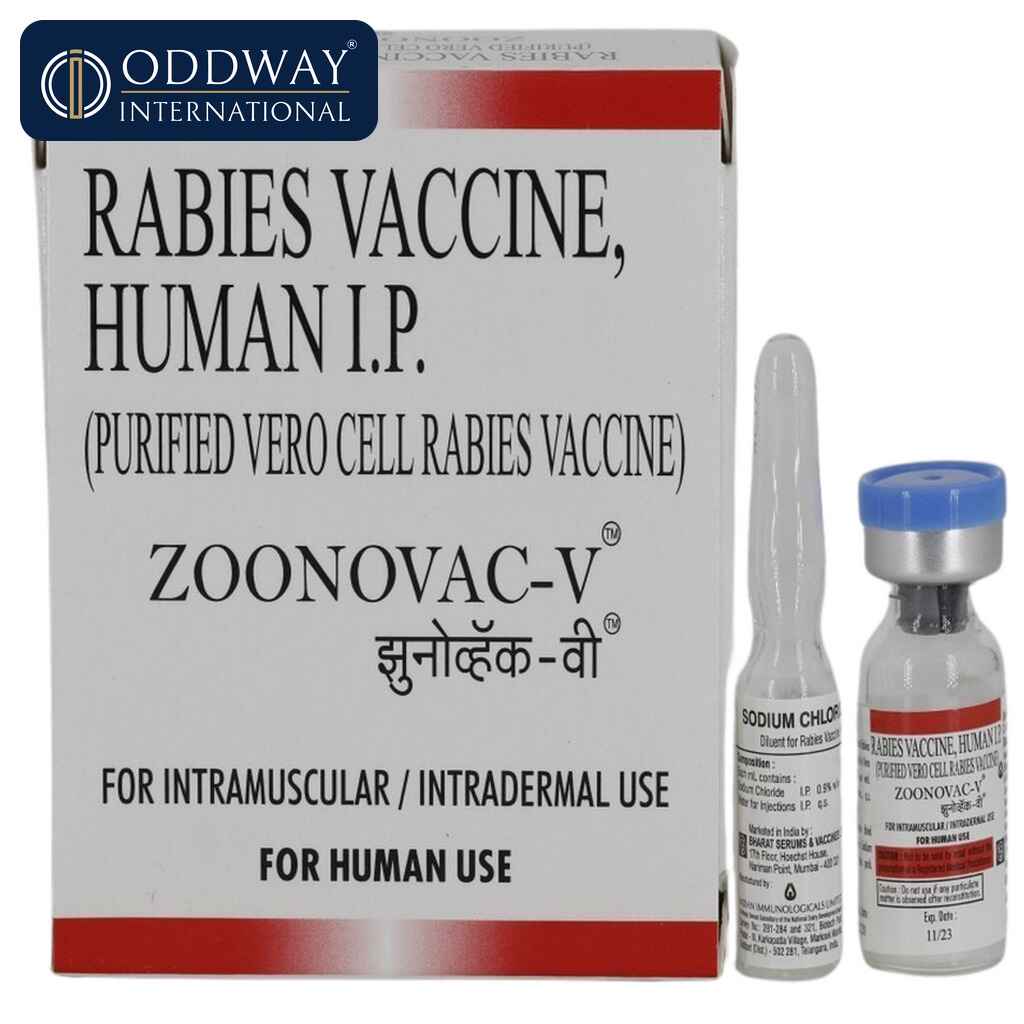
Zoonovac V Inj
Brand Name: Zoonovac V Inj
Composition: Purified Vero Cell Rabies Vaccine for Human Use
Dosage Form: Injection
Packaging: 1 Vial
Manufacturer: Bharat Serums & Vaccines Ltd
Send Enquiry for Zoonovac V Inj
Product overview for Zoonovac V
Source Zoonovac V for institutional procurement where permitted and subject to local regulations. Oddway International supports B2B buyers that require consistent documentation, controlled handling, and supply-ready coordination for Zoonovac Inj shipments. Moreover, we align supply planning with your tender timelines, hospital schedules, or distributor replenishment cycles to reduce operational delays.
Zoonovac V is requested by healthcare and public health channels that manage immunization programs and emergency preparedness. Consequently, procurement teams often prioritize batch traceability, validated cold-chain practices, and clear labeling for cross-border movement. We coordinate these requirements at the order stage to help you avoid avoidable rework during import clearance.
For buyers consolidating multiple product categories, we can integrate generic medicine sourcing into the same supply workflow. Additionally, if your portfolio includes rabies immunization products, we can support category-level procurement through Anti Rabies Vaccine wholesaler channel planning, subject to availability and destination rules.
Therapeutic category/Drug classification
Zoonovac V falls under vaccines/immunologicals and is positioned as a rabies vaccine product line in trade discussions. The keyword reference zoonovac v rabies vaccine is used for identification in procurement documents and comparative sourcing. However, final classification, indications, and immunization schedules must always follow the approved label in the destination country and the prescribing authority’s protocol.
Typical institutional use cases include stocking for immunization services and emergency response programs, where permitted. Potential adverse reactions and safety information vary by product labeling and local guidance; therefore, buyers should review the pack insert, storage requirements, and contraindication statements before distribution or administration. We provide batch-specific paperwork and can share available safety documentation such as SDS/MSDS upon request, subject to manufacturer availability.
Zoonovac V Inj pricing and minimum order quantity
For Zoonovac V price requests, we quote based on strength, pack configuration, requested brand/manufacturer options, destination country, and shipment mode (air/sea). Additionally, pricing may vary by batch availability, lead time, and cold-chain requirements. We do not publish fixed rates; instead, we provide a formal proforma invoice for procurement approval.
Minimum order quantity (MOQ) depends on packaging, carton configuration, and the feasibility of maintaining required temperature conditions throughout transit. Consequently, buyers should share target quantity, delivery timeline, and import license details (where applicable) so we can confirm MOQ and lead time accurately.
Available strengths and packaging for Zoonovac Inj
Zoonovac Inj is supplied in suitable presentations subject to availability and requested brand. Strength and pack details can differ by market authorization and manufacturer. Therefore, we confirm the exact strength, vial/syringe presentation, and pack size during quotation to match your tender or hospital formulary requirement.
- Strength: as per approved label and batch availability
- Dosage form: injection (vaccine)
- Packaging: as per manufacturer pack (confirmed at order stage)
- Storage: temperature-controlled handling as required by label
Wholesale supply and named patient supply capabilities with Zoonovac V supplier support
Oddway International operates as a Zoonovac V supplier for wholesale orders, supporting regulated and semi-regulated markets. Moreover, we assist importers, hospital procurement teams, NGOs, and distributors with order planning, batch reservation (where possible), and shipment scheduling aligned to your receiving capacity.
For urgent requirements, we also support Named Patient Supply (NPS) shipments where permitted. Consequently, we can coordinate priority packing, rapid documentation, and time-sensitive dispatch options to help meet clinical deadlines, while ensuring compliance with destination-country rules.
Quality assurance and global standards
We follow GDP-aligned handling practices and work with manufacturers and logistics partners to maintain product integrity in transit. Additionally, we support documentation commonly requested in local trade, such as Certificate of Analysis (COA) when available, batch details, and shipping temperature guidance. However, all supply remains subject to manufacturer release processes and local import regulations.
We emphasize traceability across the supply chain, including batch/lot recording and documentation consistency. As a result, procurement teams can reconcile received stock with purchase orders and import paperwork more efficiently.
Why source Zoonovac V Inj from Oddway International
Oddway International is a trusted Indian pharmaceutical supplier focused on B2B supply reliability. We communicate clearly on lead times, documentation readiness, and shipment constraints for temperature-sensitive products. Furthermore,
We prioritize responsive coordination across quotation, order confirmation, and dispatch. Therefore, buyers receive structured updates, and we reduce back-and-forth during critical steps such as label confirmation, batch allocation, and airway bill sharing.
Logistics and documentation Support for Zoonovac V
We arrange shipping with attention to route selection, transit time, and special handling needs, including cold-chain solutions where required. Additionally, we support standard documentation such as commercial invoice, packing list, certificate of origin (when applicable), and courier/airline paperwork. Consequently, importers can proceed with customs clearance more smoothly, subject to local authority requirements.
5 reviews for Zoonovac V Inj
Shipping Options
We offer a variety of shipping options, including Express Mail Service (EMS), USPS, DHL, FedEx, TNT, UPS, Aramex, Air Cargo, and sea freight.
Terms and Conditions
- Bulk Orders: You are responsible for any local import duties and taxes in your country.
- For Patients: When ordering prescription medicines for personal use, or for a friend or relative, a valid medical practitioner's script or prescription is required.
- Return and Refund Policy: Due to the nature of our products, we cannot accept returns or exchanges once a purchase is made. However, in the event of non-delivery, you are eligible for either a 100% refund or a reshipment of your order.
Frequently Asked Questions
Yes. We support B2B wholesale procurement of Zoonovac V Inj for, distributors, hospitals, and institutional buyers, subject to availability.
Zoonovac V price is quoted through a proforma invoice based on requested strength and packaging, quantity, destination, shipping mode, and cold-chain requirements where applicable. Share your target volume and delivery timeline for an accurate quote.
We can support NPS shipments where permitted, coordinating priority dispatch, documentation, and suitable logistics options. Requirements vary by country, so we review the case details before confirmation.
MOQ depends on manufacturer pack configuration, batch availability, and the feasibility of maintaining required handling conditions during transit. Provide your destination and required timeline so we can confirm the MOQ and lead time.
Common documents include commercial invoice, packing list, airway bill/shipping paperwork, and supporting documents such as COA or SDS/MSDS when available from the manufacturer. Final documentation depends on requirements and order specifics.
Ahmed K. –
Our main requirement was documentation consistency for internal audit. Oddway provided the files promptly and coordinated shipment steps clearly. We will reorder for future tenders if availability aligns.
Mei L. –
Good experience overall. They confirmed packaging details before invoicing and kept us informed about batch readiness. Delivery was within the window we planned for.
Luis A. –
We needed tight coordination with our freight forwarder and Oddway stayed responsive through dispatch. Documents were shared in a structured way, which reduced back-and-forth during clearance.
Rania S. –
Lead time was explained upfront and updates were consistent. A couple of label confirmation points took an extra day, but the team handled it without confusion.
Daniel M. –
Communication was quick and practical. We received clear options for shipping and the paperwork matched what our clearing agent requested.